Poster
Modeling immuno-oncology in a humanized mouse
NOD-scid IL2R?null (NSG) mice engrafted with human peripheral blood mononuclear cells (hPBMCs) have been utilized in xenograft models with success to evaluate efficacy and pharmacodynamics of immune checkpoint inhibitors targeting epitopes on human T-Lymphocytes1. This model is unique over conventional xenografts because functional human T-cells can target human epitopes on a tumor in a mouse with directly translatable clinical significance. NSG mice are deficient in dendritic and macrophage cell populations and carry two specific mutations rendering them highly deficient of lymphocytes; the Pkrdc mutation disables aspects of DNA repair that renders the mouse B- and T-cell deficient, and deletion of IL2R? blocks NK cell maturation2. This combination of deficiencies allows for the engraftment of hPBMCs and allows for the expansion of human T-lymphocytes in the NSG mouse.
The hPBMC engrafted NSG mouse does have caveats that must be evaluated prior to human donor and tumor model selection. First, administration of human immune cells to a mouse results in T-cell mediated toxicity consistent with Graft Versus Host Disease (GvHD), in which human lymphocytes attack mouse tissues, limiting the therapeutic window3. Second, engraftment efficiency, cell types present, and activity have been observed to be variable between human donor sources4. Finally, we have observed instances of hPBMC rejection of the human tumor xenograft (data not shown). Evaluation of these parameters with the donor source and tumor model of choice, prior to initiating definitive efficacy studies, is critical for the success of translational early development programs.
Experimental Design
The objective of this study is to evaluate the engraftment of human T-lymphocytes from hPBMCs administered to NSG mice bearing subcutaneous human tumor xenografts. Animal care and use was performed in conformance with the Guide for the Care and Use of Laboratory Animals in an AAALAC-accredited facility. NSG mice (Jackson Laboratories, Bar Harbor Maine, USA, strain #0005557) bearing subcutaneous MiaPaCa-2 human pancreatic ductal adenocarcinomas were intravenously administered hPBMCs from four different normal healthy donors (Hemacare, Los Angeles CA, USA). Tumor growth, bodyweight and evaluation of GvHD-like characteristics were performed. Whole blood was collected for flow cytometry analysis of human lymphocyte markers to confirm donor engraftment of CD45+ cells, including CD4+ and CD8+ cells, on days 14, 28, and 42 post hPBMC administration (Figure 1).
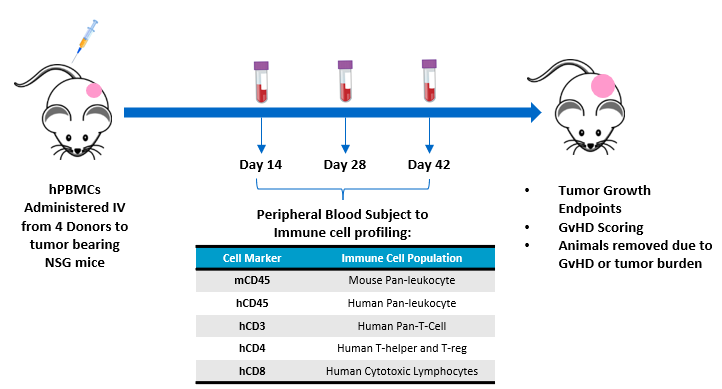
Figure 1 ? Experimental design assessing hPBMC engraftment in MiaPaca-2 tumor bearing NSG mice.
Tumor Growth, Bodyweight, and Disease Progression
MiaPaCa-2 tumor doubling time (Td) of untreated control animals was 9.1 days, Td of animals administered hPBMCs ranged from 8.7 to 11.6 days across each donor group. Variability in growth was observed across individuals administered hPBMCs beyond 30 days following implant. Tumor growth before 1000mm3 was not impacted by engraftment of hPBMCs (Figure 2). These results suggest any donors utilized would be suitable for MiaPaCa2 efficacy studies with a time to evaluation size (TES) of 1000mm3.
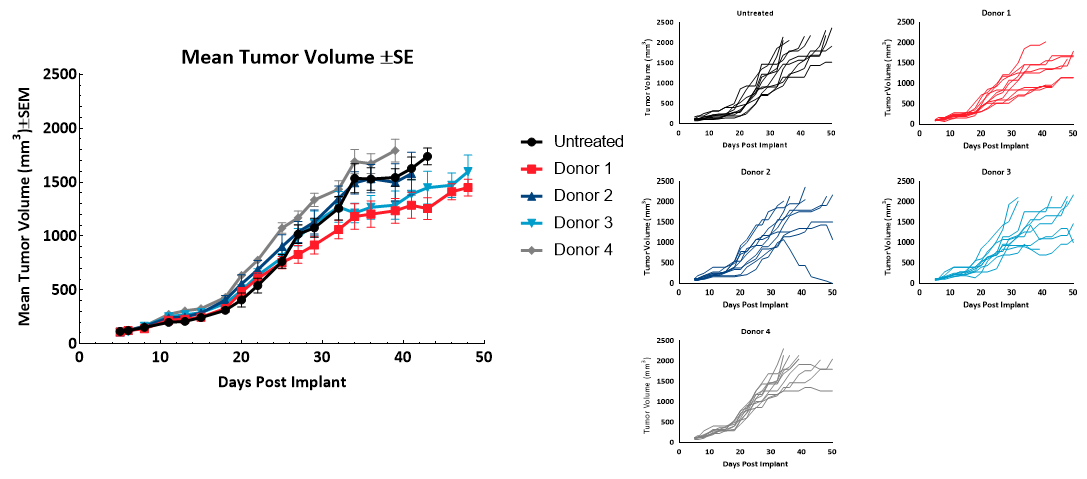
Figure 2 ? Tumor Growth of MiaPaCa2 subcutaneous tumors (n=10) following hPBMC administration.
Onset of GvHD symptoms was monitored by bodyweight and clinical observations; though not confirmed pathologically in this study, symptoms observed in this model are strongly correlated with the disease5. Symptoms include bodyweight loss (Figure 3) of greater than 10% of baseline, rough pelage, hunched posture, skin lesions, and diarrhea ? animals presenting all symptoms, or a high severity of several, were removed from study. Thirty-two days following hPBMC injection, clinical signs were observed in half or more animals of each group. Animals not removed for GvHD symptoms were removed due to a tumor burden in excess of 2000mm3, or at study termination.
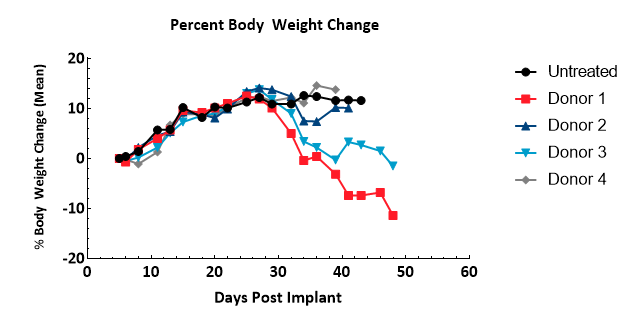
Figure 3 ? Assessment of bodyweight change following administration of hPBMCs
Engraftment and Persistence of Human Lymphocytes
Engraftment was measured 14, 28 and 42 days following hPBMC administration by immunophenotypic analysis of human immune cell markers in peripheral blood from NSG mice: mCD45 (murine pan-leukocyte), hCD45 (human pan-leukocyte), hCD3 (pan-T-Cell), hCD4 (T-helper and T-reg), and hCD8 (cytotoxic lymphocytes) were detected by flow cytometry (Figure 4).
Onset of GvHD symptoms was monitored by bodyweight and clinical observations; though not confirmed pathologically in this study, symptoms observed in this model are strongly correlated with the disease5. Symptoms include bodyweight loss (Figure 3) of greater than 10% of baseline, rough pelage, hunched posture, skin lesions, and diarrhea ? animals presenting all symptoms, or a high severity of several, were removed from study. Thirty-two days following hPBMC injection, clinical signs were observed in half or more animals of each group. Animals not removed for GvHD symptoms were removed due to a tumor burden in excess of 2000mm3, or at study termination.
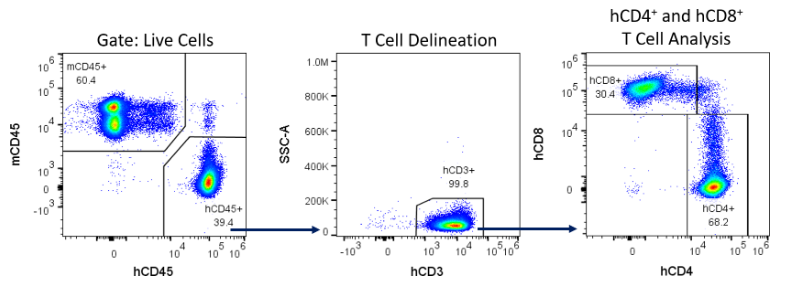
Figure 4 ? Example gating strategy for the detection of human immune cells in NSG mice reconstituted with hPBMCs.
Percent hCD45+ is calculated as the percent hCD45+ cells of total live cells (based on absolute cell counts/µL whole blood) and is used as an indicator of the extent of hPBMC engraftment. We found that %hCD45+ cells in whole blood were detected over time across all donors (Figure 5). However, Donors 2 and 4 exhibited greater increase of hCD45 cells on an individual basis compared to Donors 1 and 3 whose engraftment was not as high by day 42 post hPBMC administration. Animals across all groups achieved %hCD45 engraftment levels 28 days post hPBMC administration that is consistent with published literature on the model6.
Administration of hPBMCs to NSG mice results in persistence of human T-cells in the mouse with minimal effects on MiaPaCa-2 tumor growth. This model represents a powerful preclinical platform to examine the effects of novel agents that harness human T-lymphocytes to direct anti-tumor activity with direct clinical significance. Future work will demonstrate efficacy of FDA approved immuno-therapies against human tumor xenografts in NSG mice reconstituted with hPBMCs.
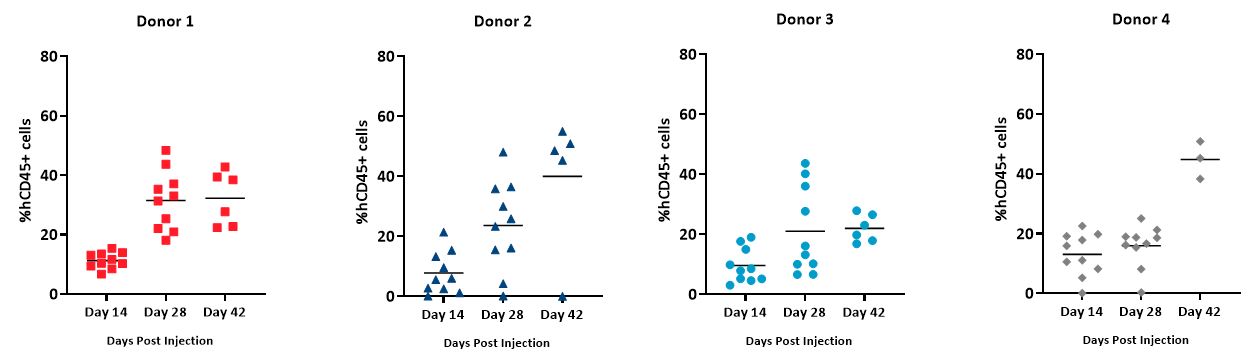
Figure 5 ? Extent of engraftment of hCD45+ cells detected in whole blood from NSG mice administered hPBMCs
Please contact our preclinical oncology scientists to see how hPBMC engrafted NSG mice can be used for your next translational immuno-oncology study.


